At the recent International Gynecologic Cancer Society Annual Global Meeting in Seoul, South Korea, Duke Cancer Institute gynecologic oncologist Angeles Alvarez Secord, MD, MHSc, presented real-word data from the DCI Endometrial Cancer Molecularly Target Therapy (EMCT2) Consortium regarding pembrolizumab and lenvatinib for endometrial cancer patients. While the FDA-approved combination of pembrolizumab and lenvatinib regimen improved survival compared to standard chemotherapy for endometrial cancer patients, the side effects are significant, said Dr. Secord, who serves as president of the Society of Gynecologic Oncology. View the poster presentation.
“The concern about tolerability of this combination has led to variation in prescribing patterns. The findings revealed that most providers initiate levantinib therapy at 14 mg daily. Toxicity was decreased with 14 mg, and lower doses with no decrement of survival outcomes and response rates were demonstrated,” said Dr. Secord.
The EMCT2 consortium is comprised of 23 members and over 2,800 patients.
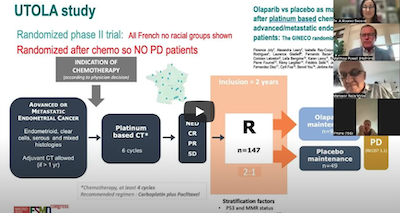
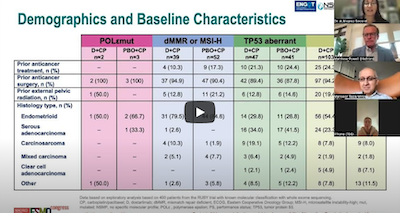
Additional clinical trial news updates on advances in gynecologic cancer treatment came from the European Society for Medical Oncology (ESMO) Congress 2023 in Spain, according to Dr. Secord.
A substantial number of gynecologic cancer practice-changing trials were presented at ESMO. Several of these trials demonstrate substantial and meaningful gains in disease control and survival outcomes for ovarian, endometrial and cervical cancer patients. Moreover, promising novel therapies with new mechanisms of action and precision targeting of cancer cells are in development. These include encouraging trial results for cervical, endometrial and ovarian cancer.
"The accelerated pace of research and active new treatments have transformed care for our patients. It’s so wonderful to see our patients with metastatic cancer responding to treatments," said Dr. Secord. "In some cases, especially with immunotherapy, we are seeing amazing responses that we hope will lead to long-term cures. These are exciting times in cancer care.”
Discussions about these developments were recently held by internationally recognized women’s cancer experts in a three-part video series focusing on the clinical trial data: